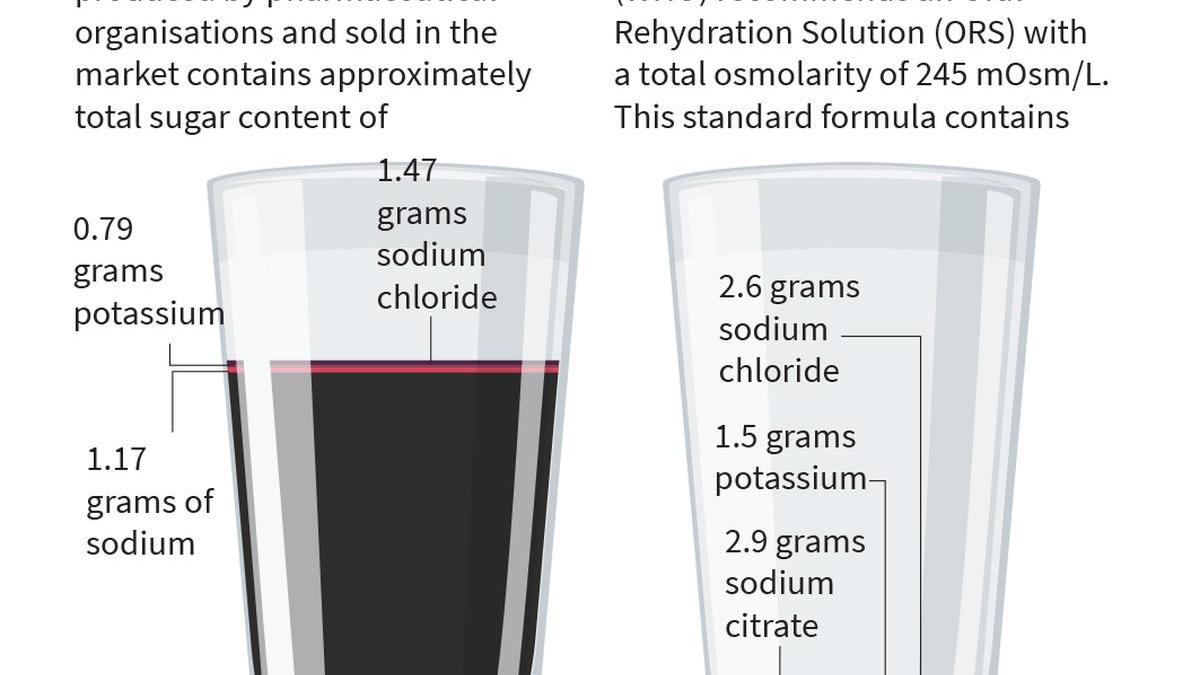
When prospects ask for ORS, pharmacies typically supply the sweetened ORSL drink as a substitute of the WHO-recommended rehydration resolution
Dr. Sivaranjani, who led the authorized marketing campaign that prompted the regulatory motion, stated the decision is a victory for India and that not a single youngster’s life ought to be endangered due to deceptive labels falsely utilizing the time period ORS. She urged the FSSAI to behave decisively to make sure full compliance and forestall such violations in future.
The judgment, delivered by Justice Sachin Datta on October 31, 2025, in Dr Reddy’s Laboratories Ltd. & Ors. vs Union of India & Anr, upheld the FSSAI’s orders dated October 14 and 15, and a subsequent enforcement communication issued on October 23, 2025. The court docket dominated that the regulator’s determination was justified on critical public well being grounds and fell squarely inside its statutory powers.
In its October orders, the FSSAI withdrew earlier approvals that had permitted using ‘ORS’ with prefixes or suffixes in product logos, clarifying that solely formulations strictly adhering to World Health Organisation (WHO) requirements could also be labelled as Oral Rehydration Salts or ‘ORS’. The ruling follows an eight-year marketing campaign by Dr. Sivaranjani, who had in 2022 filed a Public Interest Litigation (PIL) earlier than the Telangana High Court difficult the sale of drinks misleadingly marketed as ORS.
Petitioner’s problem
Hyderabad-based Dr. Reddy’s, which manufactures ‘Rebalanz Vitors’, contended that FSSAI’s determination was arbitrary, taken with out discover, listening to, or session with stakeholders. The firm claimed that the orders had a major affect on its operations and proprietary trademark rights and infringed its elementary rights.
The petition identified that giant portions of its beverage, bought in 200 ml packs beneath apple, orange, and mango flavours, had been already manufactured and distributed earlier than the FSSAI’s order, and had been now mendacity unsold. According to the petition, Dr. Reddy’s Laboratories had a complete of 8,47,181 items of its “Rebalanz Vitors” beverage mendacity unsold in its completed items stock, with a mixed worth of roughly ₹1.39 crore as of October 15.
The firm argued that with out reduction, it could undergo heavy financial losses for the reason that merchandise couldn’t be bought with present packaging.
FSSAI’s findings
During the ultimate listening to on October 31, the FSSAI, represented by Additional Solicitor General Chetan Sharma, famous that earlier approvals (granted in July 2022 and February 2024) had been conditional and topic to assessment, and may very well be withdrawn in public curiosity. It discovered that disclaimers on product labels had been ineffective when model names prominently featured ‘ORS’, utilizing comparable fonts and color schemes as medical ORS formulations.
Court’s evaluation
Justice Sachin Datta noticed that the FSSAI’s actions had been impelled by critical public well being issues and supposed to safeguard customers, significantly susceptible teams. The court docket declined to intrude with the professional physique’s technical and regulatory choices, holding that it was not applicable for judicial assessment to second-guess such coverage determinations.
“The strategy of utmost warning as adopted by the respondents on a difficulty which may endanger public well being gravely can’t be faulted with,” the court docket stated, including that statutory obligations beneath the FSS Act couldn’t be subordinated to personal industrial losses.
Relief sought and last course
The court docket recorded that Dr. Reddy’s had stopped manufacturing of recent shares and expressed willingness to relabel or rebrand present stock. The firm additionally sought permission to promote merchandise already within the provide chain to keep away from monetary loss.
The court docket declined to move any direct reduction, as a substitute directing the FSSAI to contemplate the petitioner’s illustration on this concern and move a reasoned order inside one week, after affording a chance of listening to. The writ petition was accordingly dismissed, whereas granting liberty to the petitioner to strategy the FSSAI relating to the unsold inventory.
Published – November 02, 2025 11:37 am IST








